Novel Enabling Tools and Models Supporting Development of Interventions for Enteric Dysfunction (Round 12)
Opportunity:
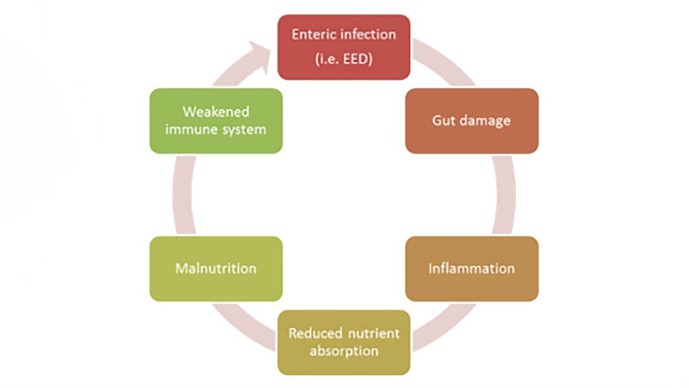
In young children in the developing world, persistent and repeated bouts of enteric infection, malnutrition, and environmental factors interact in a complex and ill-defined way to negatively impact gut health and function. These events commonly lead to both a) repeated episodes of acute secretory diarrhea (ASD), and b) impaired gut function, commonly known as environmental enteric dysfunction (EED) or tropical enteropathy. These two conditions cause significant problems for children in the developing world: ASD kills nearly 800,000 children worldwide every year, and EED causes widespread morbidity (and likely increased mortality), including stunting and impaired cognitive development. The fundamental physiology and pathology underpinning ASD and EED includes many factors: altered enteric ion channel activity, which leads to hyper-secretion of water and electrolyte imbalances; impaired nutrient absorption and transport; hormonal and neuroendocrine dysregulation; increased inflammation and resultant sequela; compromised enteric immune function and barrier function; and perturbed gut microbiome interactions. The critical inter-dependencies between these gut functions, not yet fully defined or understood, result in significant and chronic gut function decline, leading to a well-described vicious cycle as graphically shown below.
The Challenge:
Given the pernicious broad impact of ASD and EED on nutrition, growth, cognition, immune response, and overall health, particularly in children under five years of age (our target population), we are interested in developing therapeutic interventions to improve child health in the developing world by identifying agents to restore normal gut function in the target population. Importantly, given the wide variety of bacteria, viruses, and other pathogens (e.g. cryptosporidium) that infect our target population, we are interested in developing host-directed therapies rather than agents that would kill the pathogen. Unfortunately, few candidate molecules or validated therapeutic approaches are available, and the path from discovery to translation to clinical efficacy is unclear. More specifically, we lack preclinical assays, methods, tools, and models by which potential therapeutic interventions can be identified, developed, compared, and prioritized – particularly methods which can de-risk the approach as it transitions from in vitro and/or in vivo assays into human clinical trials.
What We Are Looking For:
Thus, we are soliciting novel and innovative ideas for tools, approaches, and models to support preclinical development of new agents for ASD and EED. We particularly seek approaches which recapitulate the pathophysiology of impaired gut function as it occurs in our target population. We will consider a wide variety of approaches, as outlined below, provided a case can be made for how the approach will facilitate and support the development of new host-directed therapeutic interventions. We specifically request that applicants do not submit proposals focused on the identification and/or development of new therapies, as this is outside the scope of this GCE call. Likewise, we will not consider proposals specifically focused on developing or using gut-function biomarkers, as we have already made significant investments in this area. However, please note we do distinguish between general gut function biomarkers and biological target biomarkers, and thus we will consider proposals for novel biomarkers indicative of target activation or inhibition which can be used to facilitate the development of new ASD and EED therapies. Further guidance for what would be considered in and out of scope is provided below.
A few examples of work that would be considered for funding:
- Novel or under-explored animal and/or tissue models which recapitulate the hypersecretory intestinal states which occur in humans during ASD;
- Novel "humanized" animal models containing human cells and/or intestinal segments, or novelin vitro applications of human cells and tissues;
- Animal models designed to measure target or mechanism effectiveness in a malfunctioning/damaged gut mimicking conditions found in ASD or EED;
- Tools, probes, and approaches to dynamically and accurately quantify intestinal water loss, including contributions of ion flux, neuroendocrine activation, and paracellular water transport;
- The development of novel biomarkers indicative of target activation or inhibition which might be used to facilitate clinical development of new therapies for ASD or EED;
- New models of ASD or EED which accurately capture the contribution of physiologically regulated intestinal epithelial transport processes of the GI tract and the importance of non-epithelial intestinal components, including the microbiome, neuroendocrine system, and activity of enteroendocrine cells;
- In vitro models recapitulating intestinal physiology and disease pathology which integrate multiple gut functions and processes encountered in ASD or EED, including alterations in gut motility, permeability, and transport;
- Highly innovative platforms and models which are robust, quantitative, and closely mimic human pathology for high predictive value.
We will not consider funding for:
- Applications proposing basic research not focused on the development of improved models, assays, platforms, or approaches to facilitate the development of new interventions for ASD or EED;
- Proposals focused on identifying or developing new targets, therapies, or interventions to treat or control ASD or EED, including novel small molecular weight compounds, biologics, prebiotics or probiotics, “functional foods”, and microbiomic approaches;
- Approaches or methods which are only relevant to the development of agents directed against pathogens (as opposed to the host), or would only be applicable only to a particular pathogen or condition, e.g. vaccines;
- Applications solely focused on the discovery or development of interventions for preventing ASD or EED; for example the development of vaccines, improved sanitation, microbiome replacement therapy, or improved nutrition;
- Standard well-defined and commonly used assays or models routinely used for studying ASD or EED, including:
- For ASD: standard electrophysiology screens using intestinal cell lines, Ussing chambers, rodent open- and closed loop models, and standard enterotoxin, enteropathogen or chemical (e.g. castor oil) challenge models;
- For EED: well-described animal models and well-known methods to create disease states with pathophysiological processes similar to EED (e.g. chemical or pathogen insult, genetic modified animal models; and surgical ligature or loop methods, bacterial overgrowth models);
- Proposals involving clinical trials in human volunteers or patients;
- Proposals focused on identifying or developing novel biomarkers for ASD or EED.